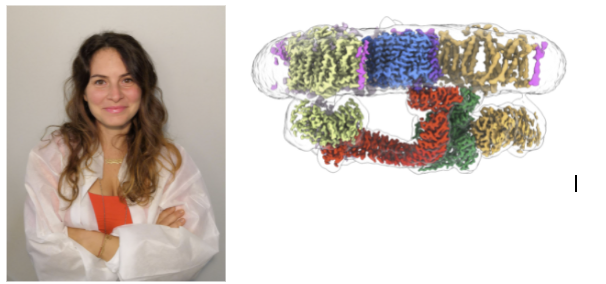
The ankyrin-1 complex tethers the spectrin-actin cytoskeleton to the red blood cell (RBC) membrane, and acts as a metabolic hub, connecting membrane proteins that are involved in gas exchange, pH control, and regulation of cellular volume and deformability. Mutations in components of the complex lead to inherited defects in erythrocyte shape and stability, such as hereditary spherocytosis. Ankyrins are also broadly expressed adaptors functioning as master-organizers of membrane-associated protein complexes in neurons and other cell types. However, the precise composition of the ankyrin-1 complex, and the structural basis for membrane association and recruitment of target membrane proteins remains unknown in any context. We solve the single particle cryo-EM structures of the human ankyrin-1 complex. Our structures reveal the architecture of this 1.2 MDa supercomplex, which includes the Rhesus polypeptides RhCE & RhAG, ankyrin 1, protein 4.2 and three copies of the dimeric band 3 anion exchanger bound to glycophorin A, assembled into a 1.2 MDa supercomplex. Additional complexes carrying one aquaporin-1 (AQP1) tetramer are also identified. The structure of membrane-bound ankyrin shows that the first five repeats adopt an unexpected T-shaped configuration whereby the inner groove is oriented parallel to the membrane, facilitating recognition of integral membrane binding partners such as RhCE and AQP1. Both the inner groove and the convex outer surface of ankyrin participate in specific interactions with protein 4.2 and band 3. Our structures also reveal the architecture and stoichiometry of the heterotrimeric Rh channel, highlighting the role of ankyrin in mediating clustering of multiple copies of structurally diverse membrane proteins.
22/04/2022 ORE 17.00 Francesca Vallese Dept. of Anesthesiology Columbia University, New York, USA